Key Points
-
This study evaluated the appropriateness of 4F-PCC orders before and after implementation of a pharmacist-led anticoagulation reversal stewardship program at a community teaching hospital and Level II Trauma Center.
-
An updated local anticoagulation reversal guideline was implemented to align with the American College of Cardiology 2020 guidance on anticoagulation reversal.
-
There was a decrease in inappropriate 4F-PCC orders, attributed to pharmacists recommending against the use of 4F-PCC for patients who did not meet guideline criteria, without affecting the efficacy and safety outcomes of the patients at the study institution.
Introduction
Anticoagulation agents, such as warfarin and direct oral anticoagulants (DOACs), are widely used for a variety of indications, such as stroke prophylaxis in atrial fibrillation and treatment of venous thromboembolism (VTE). It is estimated that over 7 million patients in the United States are treated with anticoagulants and are thus at an increased risk of bleeding and substantially increased morbidity and mortality.1 This increased prevalence of anticoagulation use is likely due to the aging population of the country.2 This increase in anticoagulation use may in turn increase the need for anticoagulation reversal. Four-factor prothrombin complex concentrates (4F-PCC) are used to reverse warfarin and Factor Xa inhibitors, but it is important to determine how recently the anticoagulant was dosed and if 4F-PCC is appropriate for each patient. If 4F-PCC is used inappropriately for anticoagulation reversal, consequences may include thrombosis leading to pulmonary embolism, stroke, myocardial infarction, or deep vein thrombosis.3
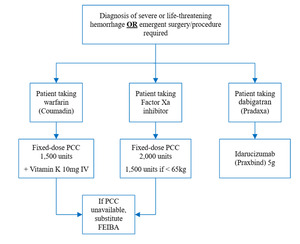
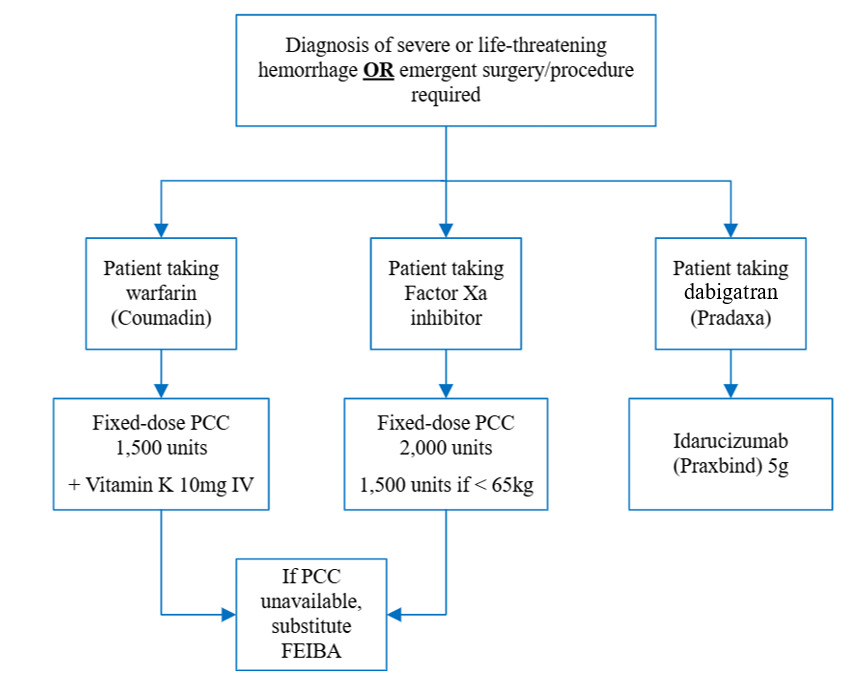
According to the American College of Cardiology (ACC), indications for anticoagulation reversal include major acute bleeding and emergent surgical intervention. Major acute bleeding is defined as bleeding at a critical site causing hemodynamic instability with or without a decrease in hemoglobin ≥2 g/dL or administration of ≥2 units of packed red blood cells (PRBC). Critical bleed sites include intracranial, spinal, intraocular, intra-abdominal, retroperitoneal, intramuscular, and intra-articular hemorrhages, pericardial tamponade, profuse epistaxis, and hemothorax.4 The ACC provides a decision pathway for anticoagulation reversal in the setting of life-threatening bleeding. In this pathway, 4F-PCC is recommended to reverse warfarin and the Factor Xa inhibitors apixaban and rivaroxaban. ACC guidance recommends administration of 5-10 mg IV Vitamin K prior to 4F-PCC for warfarin reversal. There are several studied dosing strategies that may be used for 4F-PCC, including INR-based, weight-based, and fixed-dosing options.4 The institution utilizes a fixed-dosing strategy for 4F-PCC dosing based on the anticoagulant agent (Appendix A).
This study was modeled after similar pharmacist-lead anticoagulation reversal stewardship programs. Literature review of three unique stewardship programs have included pharmacist interventions related to recommending changes in reversal agent, dose, and further laboratory monitoring. Wychowski et al. assessed the effects of implementing an anticoagulation stewardship program led by clinical pharmacy specialists in a community hospital. The program involved daily electronic health record (EHR) reviews to identify high-risk patients on anticoagulants and provide targeted recommendations related to pharmacokinetic/pharmacodynamic optimization, therapy selection, and monitoring. One of the recommendation subcategories included anticoagulation reversal with 4F-PCC, vitamin K, and protamine. Following implementation, inappropriate 4F-PCC use decreased from 55.8% to 2.6%, corresponding to a reduction in excess drug costs from $104,274 to $10,831. This stewardship initiative was associated with improved anticoagulant use and patient outcomes, as well as substantial cost savings.5 Waheed et al. reported on the development of a multidisciplinary blood factor stewardship program in response to increased off-label use of coagulation factors dispensed from the blood bank rather than the pharmacy. After blood factor dispensing shifted to pharmacy, a stewardship program was implemented by pharmacists, hematologists, surgeons, anesthesiologists, and intensivists. Utilization of 4F-PCC and recombinant factor VIIa (rFVIIa) was assessed for six months before and after program implementation. Although overall use of these products were similar in both study periods, the program resulted in a cost-savings of $101,736 within the first six months. The authors concluded that a coagulation factor stewardship program can promote more-cost effective use of hemostatic agents.6 Procopio et al. evaluated implementation of a clinical pharmacist-driven anticoagulant reversal program in the setting of suspected bleeding or need for an urgent procedure. This program was exclusively run by clinical pharmacy specialists that established collaborative guidelines with physicians. Every order for an anticoagulation reversal agent prompted a call to an on-call clinical pharmacy specialist for approval and consultation. It was reported that the primary outcome of hemostatic efficacy was achieved in 84% of patients within 24 hours, and 90% in patients who received 4F-PCC. The safety outcomes evaluated thromboembolic events within 30 days and in-hospital mortality. Nine (7.4%) patients developed thromboembolic events after administration of an anticoagulation reversal agent, and in-hospital mortality was 26%. It was determined that the interventions made by the stewardship program resulted in a total medication cost avoidance of $1,005,871.78. The authors concluded that this was the first study to evaluate the impact of a pharmacist-driven anticoagulation reversal program on hemostasis, safety, and cost outcomes in a real-world population.7
Prior interventions described in previous literature have demonstrated achievement of effective hemostasis after anticoagulation reversal, increased appropriateness of anticoagulation reversal use, and cost savings of thousands of dollars. This study aimed to implement a program at a community hospital modeled after those described in prior studies, with the goal of achieving similar outcomes. To our knowledge, no prior studies have evaluated a stewardship program in which all pharmacists at the institution were trained to participate in improving the appropriateness of 4F-PCC use.
The study institution uses a local anticoagulation reversal guideline for pharmacists and providers to guide clinical decision making for patients on anticoagulation agents requiring reversal for major acute bleeding or emergent surgical intervention. With the update in the ACC guidance in 2020, the study institution revised the local guideline with approval from the institutional pharmacy and therapeutics committee. All pharmacists and trauma surgery providers received targeted education to improve the appropriateness of 4F-PCC orders at the institution. This study evaluated the appropriateness of 4F-PCC orders, patient safety, and cost avoidance before and after implementation of a pharmacist-driven anticoagulation reversal stewardship program at a community teaching hospital and Level II Trauma Center.
Objective
The purpose of this study was to evaluate the effect of implementing a pharmacist-led anticoagulation reversal stewardship program on anticoagulation reversal appropriateness at a 373-bed community teaching hospital and Level II Trauma Center.
Methods
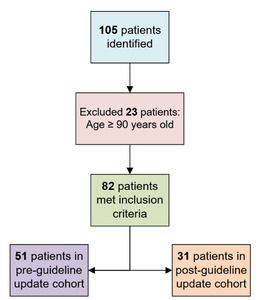
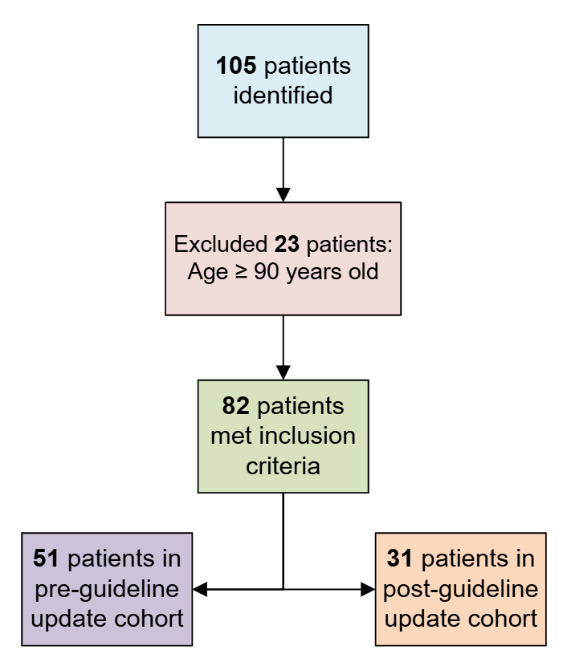
This study was a single-center retrospective cohort study. Patients were identified through a report run in the EHR which identified all orders for 4F-PCC from June 1, 2024 to March 31, 2025. This report was then reviewed by the principal investigator and patients were screened using the inclusion and exclusion criteria described below. The updated local anticoagulation reversal guideline was published after institutional pharmacy and therapeutics committee approval and education on the updated guidance began on January 9, 2025. A report was run through the hospital’s electronic health record (EHR) to identify patients. Informed consent was waived in this investigation and only the study investigators had access to protected health information.
Order verification for patients ordered 4F-PCC was performed by any hospital pharmacist at the institution. Order verification review occurred 24/7 with 4F-PCC treated as a STAT order within the verification queue which may be reviewed by a centralized or decentralized clinical or general pharmacist, depending on the order time and patient’s location within the hospital. Clinical pharmacy specialists are only available Monday-Friday during dayshift at the hospital but were available on-call if needed for clinical support. During both the pre- and post-guideline periods, pharmacists had access to a decision support resource in the form of an institutional anticoagulation reversal policy available on the hospital’s intranet. Providers ordering 4F-PCC had access to clinical decision support tools, including an EPIC-based anticoagulation reversal order set with an integrated checklist pathway for each anticoagulant. Providers also had access to the institutional policy to guide appropriate anticoagulation reversal.
Education on the updated local anticoagulation reversal guideline and accompanying pharmacist intervention documentation template was delivered as an in-service led by the principal investigator during a monthly pharmacy staff meeting. Both physical and electronic versions of the educational materials were distributed and made available to all staff pharmacists and resident pharmacists. The trauma surgery service received targeted education through two live presentations by the principal investigator, outlining the updated guideline and pharmacist intervention process. Additionally, an educational tool addressing the use of 4F-PCC and vitamin K in cases of supratherapeutic INR secondary to cirrhosis without documented warfarin or Factor Xa inhibitor use was developed and electronically distributed to all pharmacy staff and residents. Education efforts began concurrently with the implementation of the updated guideline on January 9, 2025, and were reinforced periodically through March 31, 2025. Based on historical institutional trends indicating that trauma surgery providers were the primary prescribers of 4F-PCC, educational outreach for providers during this period was focused on this service.
Inclusion and exclusion criteria
Patients were included if they were adults aged 18-89 years and ordered 4F-PCC during the study period. Patients were excluded if they were ≥90 years old, pregnant, or incarcerated.
Primary endpoint
The primary endpoint was 4F-PCC appropriateness based on indication and dose. Appropriate indications for anticoagulation reversal included major acute bleeding at a critical site (i.e., intracranial, spinal, intraocular, retroperitoneal, intraarticular, pericardial, or intramuscular with compartment syndrome), bleeding causing a decrease in hemoglobin by ≥2 g/dL or leading to transfusion of ≥2 units of whole blood or PRBC, or bleeding causing hemodynamic instability (i.e., HR ≥100 bpm, SBP <90 mmHg, or decrease in SBP ≥40 mmHg) related to known or presumed anticoagulation use within 48 hours. Orders for anticoagulation reversal for emergent surgical intervention were deemed appropriate if the surgery was a high-risk bleeding procedure or the surgery occurred within 3-4 hours of reversal. High-risk bleeding procedures were defined based on definitions derived from the ACC, which include major cardiovascular surgery, neurosurgery, major orthopedic surgery, major abdominopelvic and thoracic surgery, and other major cancer or reconstructive surgery.5 Appropriate doses were based on the local guideline recommendations for fixed dosing based on anticoagulation agent (Appendix A).
In this analysis, the composite endpoint was constructed by combining multiple clinically relevant outcomes into a single measure to comprehensively assess appropriateness of 4F-PCC orders at the institution. Each component was weighted equally, where all outcomes contributed the same degree of influence to the overall outcome.
Secondary outcomes
Secondary outcomes included the appropriateness of Vitamin K use, incidence of TEG studies, hospital length of stay (LOS), in-hospital mortality, and cost-avoidance. Vitamin K orders were considered appropriate based on anticoagulation being reversed (i.e., warfarin or other) and dose. Appropriate orders were those being used for complete warfarin reversal. Orders were deemed inappropriate if Vitamin K was omitted for warfarin reversal and if Vitamin K was given for Factor Xa inhibitor reversal without evidence from TEG to support its use. Cost avoidance was determined by calculating the number of units avoided due to pharmacist intervention (i.e., pharmacist recommended dose decrease based on weight <65 kg for Factor Xa inhibitor reversal, etc.) multiplied by the institution’s average wholesale acquisition cost for 1 unit of 4F-PCC.
Safety outcomes
Safety outcomes included hemostatic efficacy and thrombotic events. Hemostatic efficacy was defined as re-bleeding or bleed expansion after an initial dose of 4F-PCC was provided through hospital discharge. Patients were determined to have re-bleeding or bleed expansion via chart review of patient imaging scans or provider notes. Thrombotic events were assessed 24 hours after 4F-PCC administration via chart review of patient imaging and provider notes.
Data analysis
For the primary endpoint, categorical variables were evaluated using Fisher’s exact test. For baseline patient characteristics and secondary outcomes, descriptive statistics were evaluated using median and interquartile range (IQR).
Results
Study population
The EHR report of 4F-PCC orders yielded 105 patients from June 1, 2024 and March 31, 2025. Of the 105 patients, 23 met exclusion criteria for age ≥90 years, yielding a final sample size of 82 patients with 51 enrolled in the pre-cohort and 31 enrolled in the post-cohort. There was no observed difference between median patient age (74 years vs. 76 years), anticoagulation, or indications for anticoagulation between groups. There were more male than female patients in the pre-cohort group (69% vs. 52%). Apixaban was the most prevalent anticoagulation agent being reversed (72%), with atrial fibrillation being the most common indication for anticoagulation (74%). Renal function was evaluated to determine if patients were on appropriate renally adjusted doses of Factor Xa inhibitors, if applicable. INR and time from last dose were collected to analyze the need for reversal for warfarin and Factor Xa inhibitor, respectively. Most orders for 4F-PCC at the study institution were placed by hospitalists or critical care providers (70%) while trauma surgery providers had fewer 4F-PCC orders (30%). The baseline characteristics are provided in Table 1.
Patients met criteria for anticoagulation reversal if they had a guideline-supported indication as evidenced by at least one of the following: bleeding at a critical site, transfusion of ≥2 units of PRBC, hemodynamic instability, or emergent surgery. There was a numerical difference in the site of major bleeding between cohorts, with gastrointestinal bleeding observed more in the pre-cohort than in the post-cohort group. Other bleeding sites included genitourinary, surgical site, epistaxis, and arteriovenous fistula. Vasopressor requirements, initiations of massive transfusion protocol, and emergent surgical interventions were similar between groups. Additional details regarding specific emergent procedures sites are listed in Table 2. Other procedure sites included pelvic packing, exploratory laparotomy, and orthopedic procedures.
Primary outcome
Appropriate 4F-PCC orders were defined as a composite of indication criteria present, and dosing based on the anticoagulant being reversed, as stated in the policy update. There were 10 patients in the pre-cohort, and 2 patients in the post-cohort that had inappropriate 4F-PCC orders (20% vs. 6%, P = 0.12). There was some overlap in reasons for inappropriate orders as some patients had both an inappropriate indication for reversal in addition to an incorrect dose. All instances of inappropriate orders are listed in Table 3 to demonstrate the trends observed. The most common reason for inappropriate ordering was not meeting the policy criterion for major bleeding. Non-emergent surgeries were defined as those not entering the operating room within four hours of 4F-PCC dosing or low-risk bleeding procedures. Low-risk bleeding procedures encountered included surgical incision and drainage, hemorrhoid clipping, and flexible sigmoidoscopy.
While an initial sample size calculation was not performed due to the retrospective nature of the study, a post-hoc analysis was performed to assess the likelihood that the study was able to detect the observed difference between the pre- and post-cohorts. With sample sizes of 51 patients in the pre-cohort, 31 patients in the post-cohort, and an observed P-value of 0.12, the estimated power to detect the difference was approximately 34%, indicating that the study was underpowered. Based on the observed effect size, a total of approximately 194 patients (97 in each group) would have been required to achieve 80% power at a two-sided alpha of 0.05.
Secondary outcomes
Vitamin K orders were considered appropriate if it was administered to reverse warfarin or if patients had a prolonged reaction time on TEG and if the dose was 5-10 mg administered via the IV route. There were 14 patients ordered Vitamin K in the pre-cohort, and 7 patients with orders in the post-cohort. There were 10 inappropriate Vitamin K orders in the pre-cohort, and 5 inappropriate orders in the post-cohort (71% vs. 71%, P = 1.0). As stated previously, there was some overlap for reasons for inappropriate Vitamin K orders as some patients may have met multiple inappropriate criteria, though they were counted only once if this occurred. All instances of inappropriate Vitamin K orders are listed in Table 4.
Other secondary outcomes included incidence of TEG studies, mean hospital LOS, in-hospital mortality, and acquisition cost avoided. TEG studies, while underutilized at the institution, are typically used in trauma surgery or critical care patient populations to assess coagulopathies. It can be used to guide the use and dosing of 4F-PCC prior to administration, as well as post-administration to confirm correction of coagulopathy.
Acquisition cost was calculated by multiplying the units avoided by the average wholesale price per unit of 4F-PCC ($1.30). Units avoided was calculated as the sum of 4F-PCC units not given to a patient because of an appropriate pharmacist intervention recommending withholding reversal therapy. The average wholesale price per unit figure was calculated from three orderable products found on the wholesaler electronic ordering platform, in which prices may vary per day. Table 5 demonstrates the breakdown of secondary outcomes between cohorts.
The pharmacists evaluating 4F-PCC orders documented the performance of a targeted medication reconciliation and/or patient chart review 6 times in the pre-cohort and 27 times in the post-cohort (12% vs. 87%, P <0.001). Pharmacists intervened on 5 orders in the pre-cohort and 12 orders in the post-cohort. Pharmacist interventions were communicated to ordering providers via secure EHR messaging, telephone calls, or in-person discussion depending on clinical urgency and provider availability. Examples of interventions the pharmacists made included recommending withholding 4F-PCC, adding Vitamin K for warfarin reversal, and withholding Vitamin K for Factor Xa inhibitor reversals. Recommendations to withhold 4F-PCC were encouraged if anticoagulation had been held for ≥48 hours, in the setting of non-life-threatening or non-emergent indications (i.e. epistaxis, gastrointestinal bleeding, etc. without hemodynamic instability or drop in hemoglobin ≥2 g/dL). Other interventions included targeted medication history or recommending TEG studies if an unknown time existed from the last anticoagulant dose.
Safety outcomes
Safety outcomes included hemostatic efficacy and thrombotic events. The rates of each safety outcome are demonstrated in Table 7. There were no thrombotic events reported in either group through patients’ hospital length of stay after anticoagulation reversal.
One patient with bleed expansion in the pre-cohort received 4F-PCC without meeting the major bleeding criterion per the guideline. Two patients in the pre-cohort that experienced re-bleeding or bleed expansion also experienced in-hospital mortality. There was no overlap between patients within these outcomes.
The guideline allows for repeat doses of 500 units, which were not formally evaluated as a safety outcome in this study. However, two patients received repeat doses that were deemed appropriate based on the same criteria required for reversal (i.e., dose, bleeding at a critical site, hemodynamic instability, transfused ≥2 units PRBC, etc.).
Discussion
Previous literature has shown that pharmacist-driven anticoagulation stewardship programs can improve hemostatic outcomes, enhance the appropriateness of reversal agent use, and generate substantial cost-savings. This report describes a stewardship program that focused on engaging all pharmacists in optimizing 4F-PCC appropriateness at the study institution. Many of the outcomes evaluated in this study were modeled after those in previous research.5–7 Outcomes such as 4F-PCC appropriateness, hemostatic efficacy, thrombotic events, hospital LOS, in-hospital mortality, and cost avoidance have been demonstrated to provide a comprehensive understanding of both clinical and operational impact. Current literature predominantly emphasizes the role of clinical pharmacists in the development and implementation of anticoagulation stewardship programs, often through consultation and approval services or updates to existing clinical guidelines.5–7 While this study also implemented an updated clinical guideline, it was accompanied by comprehensive education for the entire pharmacist staff at the institution. Additionally, trauma surgery providers received education about the protocol as previous trends indicated that trauma surgery providers were the primary prescribers of 4F-PCC at the institution. By aligning the outcome measures and implementation strategies with those previously reported, this study allows for comparison of results across different practice settings.
There was an increase in appropriate 4F-PCC orders between the pre- and post-cohort due to a non-significant decrease in inappropriate 4F-PCC orders (10 [20%] vs. 2 [6%] P = 0.12). This is attributed to documented pharmacist interventions that resulted in appropriately decreased 4F-PCC use. Pharmacists also significantly increased their documentation of 4F-PCC order analysis (6 [12%] vs. 27 [87%]). Secondary outcomes were not significantly different between groups. The guideline update did not influence the appropriateness of Vitamin K orders nor safety outcomes in patients at the institution.
While the implementation of this project did not achieve statistical significance or cost savings seen in previous literature, the trends suggest a meaningful reduction in inappropriate 4F-PCC use and highlight the potential impact of targeted stewardship interventions. This project demonstrates a need for more education for pharmacists and providers on anticoagulation reversal appropriateness. Increased familiarity and exposure to the guideline update would yield increased adherence to its evidence-based recommendations in provider orders as well as pharmacist review upon verification for reversal appropriateness. Although pharmacist clinical review increased significantly with an 87% adherence rate for documentation after the guideline was updated. Cost avoidance was less substantial because Andexanet alfa is not available at the study institution, as avoidance of its inappropriate use in previous literature would likely have a much greater financial impact.
Patients who are not receiving anticoagulation therapy but are ordered 4F-PCC represent a clinically complex subgroup, often characterized by underlying coagulopathies such as cirrhosis or acute hepatic dysfunction. In these cases, the decision to administer 4F-PCC is challenging because the hemostatic imbalance does not necessarily reflect a simple deficiency of clotting factors but rather a fragile hemostatic state. The use of 4F-PCC in this context may theoretically exacerbate thrombotic risk without providing clear benefit.8,9 An education tool for pharmacists was developed to address these scenarios by explicitly defining appropriate indications for 4F-PCC use and discouraging its use in patients with elevated INR values solely due to liver disease coagulopathies, emphasizing the potential harms and alternative management strategies. Bleeding during cardiac surgery was not included as an appropriate indication for 4F-PCC in this analysis, as historical practice at the institution did not involve its use for this purpose and patient review confirmed no cases during the study period. However, an order set for this indication does exist, and use in this setting would not be considered inappropriate, as practice patterns may vary across institutions. Referral of these cases to a clinical pharmacy specialist was encouraged to ensure alternate therapy options were discussed with the primary team for appropriate management. It is worth considering the use of TEG as an alternative recommendation for coagulopathic patients not on anticoagulation to better guide treatment decisions. In this study there were observed orders for 4F-PCC for patients not on anticoagulant agents. Education was targeted to pharmacist and trauma surgery providers; however, it was found most patients ordered 4F-PCC were from providers from other service lines at the study institution.
Limitations
This was a retrospective pre-cohort and post-cohort chart review, thus availability and accuracy of data collected may be limited due to documentation. This was a short study period with a small sample size, which was not powered to detect statistical significance. The post-hoc analysis findings suggest that the non-significant primary outcome may be attributable to insufficient sample size rather than the absence of a true difference between pre- and post-stewardship program implementation. This highlights the need for larger studies to more definitively evaluate the impact of a similar pharmacist-driven program.
Further regarding the availability of data, pharmacists on order verification of 4F-PCC would have difficulty assessing patients that did not have an accurate medication history as their appropriateness for reversal would be unknown. This might also apply to patients who receive samples or coupons for Factor Xa inhibitors, pay cash for their medications, use a pharmacy not participating in data sharing within the EHR (such as Veterans Affairs programs), or present from a facility without a medication list. It would be unclear whether anticoagulation reversal was appropriate for these patients, requiring more information gathering than time may allow due to the emergent need for medication administration. Patients not on anticoagulant medication but presenting with coagulopathies (i.e., due to cirrhosis, thrombocytopenia, uremia, etc.) and given 4F-PCC were also captured in this study as well as intra- and post-operative cardiac surgery patients. This internal guideline did not address these subsets of patients, which posed some difficulty for pharmacists to assess appropriateness for the reversal agent.
Only a single provider group was targeted for education in this initiative, as the live educational sessions were delivered exclusively by the principal investigator. Time constraints and the limited availability of the principal investigator restricted the ability to offer sessions to additional prescriber groups. Provider education was limited to the trauma surgery service based on historical institutional trends indicating that these providers were the primary prescribers of 4F-PCC. This may have limited the overall reach and impact of the guideline update on prescribing practices. By excluding hospitalists and critical care physicians from the education sessions, these providers were only made aware of the guideline update if pharmacists encountered their orders for 4F-PCC, if they attended pharmacy and therapeutics committee where approval of the guideline update was presented for voting, or if they were to seek guidance from the electronic version of the guideline available on the institution’s intranet. It was observed that most 4F-PCC orders were placed by the provider populations that did not receive education. This may be attributable to the hospital expanding neurosurgical services during the post-implementation period which resulted in intensivists taking on a higher volume of anticoagulation reversal orders than in the past. Perhaps this was also a result of the education for trauma surgery providers leading to a decrease in inappropriate orders. Future studies should expand educational interventions to encompass all prescriber groups to maximize the impact on the appropriateness of anticoagulation reversal prescribing.
Although baseline characteristics were similar between pre- and post-implementation groups, the analysis could be strengthened by employing additional statistical methods that account for temporal trends and potential confounders. An interrupted time series analysis would allow for evaluation of changes in prescribing patterns or outcomes over multiple time points, providing a more robust assessment of the intervention’s effect beyond simple pre- and post- comparisons.
Another limitation includes utilization of acquisition cost of 4F-PCC vials to calculate cost avoidance rather than true hospital cost. Acquisition cost does not capture the full economic burden associated with product use, such as storage, preparation, or administration. Pricing for 4F-PCC can also vary substantially across institutions due to differences in contracting terms, purchasing volume, and negotiated discounts. Retrospective cost calculations also have inherent limitations including the completeness of administration documentations, particularly for agents billed by activity units when the exact administered dose is not consistently recorded. This can lead to imprecise cost estimates and potential misrepresentation of true financial impact, limiting the generalizability of cost comparisons.
With regards to the safety endpoints, there was limited follow-up, specifically for thrombotic events due to the short timeframe of the study. Limiting the assessment of thrombotic complications to the period of hospitalization likely underestimates the true incidence of thrombotic events. Thrombotic complications including VTE and ischemic stroke may occur days to weeks after discharge, particularly among patients with ongoing prothrombotic risk or recent exposure to reversal agents such as 4F-PCC.10 This was chosen as a safety endpoint but with the ability to only follow patients through their encounter, it is possible these events may have occurred but were unable to be captured after hospital discharge. Extending surveillance beyond discharge would be essential to capture delayed events and more accurately characterize the long-term safety profile of anticoagulation reversal.
The generalizability of the results is limited due to the study setting being a single-center, suburban community hospital, and Level II trauma center. Large academic medical centers with multidisciplinary teams and dedicated clinical pharmacists may implement programs more readily, whereas smaller hospitals may face challenges with limited clinical pharmacy staff, dissemination and education on updated clinical guidance, and openness to pharmacist-driven interventions. Successful replication of an anticoagulation stewardship program at other institutions would require adequate pharmacy staffing, collaborative guideline development with clinicians, structured staff education, and integration within the EHR for real-time clinical decision support.
Conclusion
The implementation and education on the updated evidence-based guideline resulted in a non-significant decrease in inappropriate 4F-PCC orders. It did not influence the appropriateness of Vitamin K orders nor affect hemostatic efficacy or thrombotic events in patients at the study institution. With the rejection of five inappropriate 4F-PCC orders over the three-month post-cohort timeframe, a projected annual cost avoidance of $48,000 was observed.
Disclosures
The authors have declared no potential conflicts of interest.